Background: Richter transformation (RT) is a rare but serious transformation of chronic lymphocytic leukemia (CLL) into an aggressive lymphoma, most commonly large B-cell lymphoma (LBCL). There is currently no standard therapy for RT lymphoma and median overall survival (OS) is estimated to be only 8-12 months. Tisagenlecleucel is an autologous CD19-directed chimeric antigen receptor (CAR)-T cell therapy approved for the treatment of r/r LBCL in patients who have received ≥2 lines of prior therapy. Here, we describe the efficacy and safety of tisagenlecleucel in patients with RT in a real-world setting with a median follow-up (infusion to data cutoff) of 31 months.
Methods: Data were collected as part of a noninterventional, prospective, longitudinal study using the CIBMTR registry. All patients were treated in the United States, Canada, or Israel. Efficacy outcomes analyzed included overall response rate (ORR), progression-free survival (PFS), and OS. Safety outcomes included the incidence and severity of cytokine release syndrome (CRS) and immune effector cell-associated neurotoxicity syndrome (ICANS).
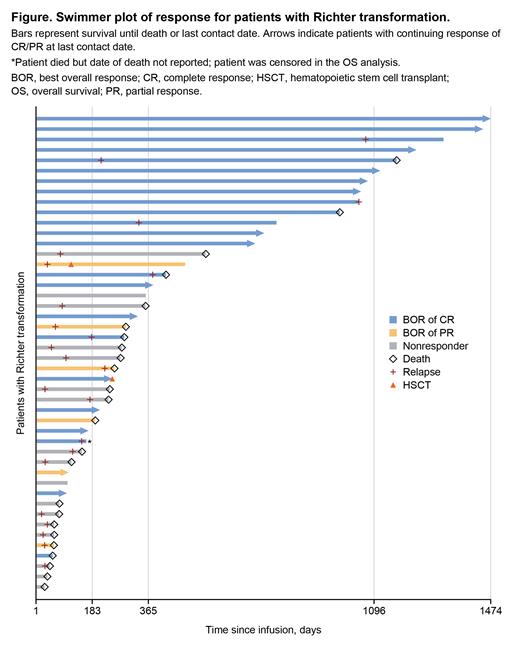
Results: As of May 4, 2023, 48 patients with RT received a single tisagenlecleucel infusion. The median age at infusion was 68 years (range, 39-83). Fifty-eight percent of patients were ≥65 years old. The median number of all prior lines of therapy was 3 (range, 1-8); 42% of patients received ibrutinib as a prior therapy. No information confirming clonal relation of LBCL to CLL in evaluated patients was available. Fifty percent of patients had primary refractory disease. Prior to infusion, 46% of patients were reported to have elevated lactate dehydrogenase (LDH). Two patients who received tisagenlecleucel infusion, and were alive at data cutoff, had not yet reached 100 days post-infusion and were therefore not evaluable for efficacy and safety. Among the 46 patients evaluated for efficacy and safety, the ORR (complete response [CR] + partial response) was 63% (95% CI, 47.5-76.8) and CR rate was 50% (n=23). In total, 23 patients (23/46, 50%) experienced disease relapse, 17 within 6 months of infusion (Figure). Twelve- and 24-month PFS were 43% and 39%, respectively; 12- and 24-month OS rates were 51% and 44%, respectively. Among 23 patients with a best overall response of CR, 57% remained in remission at data cutoff. The most common adverse events of any grade were CRS (78%), hypogammaglobinemia (46%), and ICANS (35%). Grade 3/4 CRS was reported in 11% of patients. No patient experienced grade 5 CRS. CRS was managed with tocilizumab in 81% of cases. Grade 3 ICANS was reported in 9% of patients. No grade 4/5 ICANS events were observed. Two patients went on to receive a stem cell transplant following tisagenlecleucel infusion. In total, 25 deaths were reported. The most common cause of death was progressive disease (13/48, 27%). Twelve patients (12/48, 25%) died in the absence of disease progression. Causes of non-relapse-related mortality included infection (including COVID-19, n=5), respiratory failure (n=2), malignant neoplasm (n=2), prior malignancy (n=1), multiorgan dysfunction (n=1), and severe veno-occlusive disease (n=1).
Conclusions: In the largest cohort of patients with RT receiving treatment in a real-world setting, tisagenlecleucel resulted in a high overall response rate and durable survival. The incidences of CRS and ICANS were similar to other real-world reports of tisagenlecleucel for patients with LBCL. Overall, these findings demonstrate that tisagenlecleucel could be considered a potential treatment option for patients with RT who have a high unmet medical need.
Disclosures
Shadman:AbbVie, Genentech, AstraZeneca, Pharmacyclics, BeiGene, BMS, MorphoSys/Incyte, Kite, Eli Lilly, Genmab, Mustang Bio, Regeneron, ADC therapeutics, Fate Therapeutics, Janssen and MEI Pharma: Consultancy; Mustang Bio, BMS, Pharmacyclics, Genentech, AbbVie, TG Therapeutics, BeiGene, AstraZeneca, Genmab, MorphoSys/Incyte, Vincerx: Research Funding. Frigault:Arcellx: Consultancy, Research Funding; BMS: Consultancy; Iovance: Consultancy; Kite: Consultancy, Research Funding; Novartis: Consultancy, Research Funding. Foley:Novartis: Speakers Bureau; Gilead: Speakers Bureau; Janssen: Speakers Bureau. Hill:BeiGene: Consultancy; Bristol Myers Squibb: Consultancy; Genentech: Consultancy, Other: Advisory board, Research Funding; Kite, a Gilead Company: Consultancy, Honoraria, Other: travel support, Research Funding; Pharmacyclics: Consultancy, Other: Advisory board, Research Funding; Incyte: Consultancy; Gilead: Other: Advisory board; AstraZeneca: Consultancy; AbbVie: Consultancy, Other: Advisory board, Research Funding. Jacobson:Synthekine: Consultancy, Honoraria; Sana: Consultancy, Honoraria; Novartis: Consultancy; Morphosys: Consultancy; Kite/Gilead: Consultancy, Honoraria, Research Funding; Ipsen: Membership on an entity's Board of Directors or advisory committees; Instil Bio: Consultancy; ImmPACT Bio: Consultancy; Daiichi-Sankyo: Consultancy; Caribou Bio: Consultancy; BMS/Celgene: Consultancy; AstraZeneca: Consultancy; ADC Therapeutics: Consultancy; Abintus Bio: Membership on an entity's Board of Directors or advisory committees; Abbvie: Consultancy. Jaglowski:CRISPR: Consultancy; Takeda: Consultancy; Bristol-Myers Squibb: Consultancy, Research Funding; Novartis: Consultancy, Research Funding; Kite. a Gilead Company: Consultancy, Research Funding; Caribou: Research Funding. Locke:Allogene: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other: Institutional ; Clinical Care Options Oncology: Other; Imedex: Other; Leukemia and Lymphoma Society: Other; Wugen: Consultancy, Membership on an entity's Board of Directors or advisory committees; Umoja: Consultancy, Membership on an entity's Board of Directors or advisory committees; Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other: Institutional , Research Funding; Iovance: Consultancy, Membership on an entity's Board of Directors or advisory committees; Kite, a Gilead Company: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other: Institutional , Research Funding; CERo Therapeutics: Other: (Institutional); National Cancer Institute: Other; GammaDelta Therapeutics: Consultancy; Sana: Consultancy, Membership on an entity's Board of Directors or advisory committees; Janssen: Consultancy, Membership on an entity's Board of Directors or advisory committees; Takeda: Consultancy, Membership on an entity's Board of Directors or advisory committees; Society for Immunotherapy of Cancer: Other; Aptitude Health: Other: Travel Support; BioPharma Communications CARE Education: Other: Institutional; ASH: Other: Travel Support; Pfizer: Membership on an entity's Board of Directors or advisory committees; Daiichi Sankyo: Consultancy; Amgen: Consultancy, Membership on an entity's Board of Directors or advisory committees; Bluebird Bio: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other: Institutional; Legend Biotech: Consultancy, Membership on an entity's Board of Directors or advisory committees; Emerging Therapy Solutions: Consultancy, Other; Calibr: Consultancy; Cowen: Consultancy; Cellular Medicine Group: Consultancy; Caribou: Consultancy; Bristol Myers Squibb/ Celgene: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other: Institutional , Research Funding; EcoR1: Consultancy; Gerson Lehrman Group (GLG): Consultancy; A2 Biotherapeutics: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other: Travel support. Landsburg:Karyopharm: Membership on an entity's Board of Directors or advisory committees; Curis: Research Funding; Calithera: Membership on an entity's Board of Directors or advisory committees, Research Funding; Epizyme: Membership on an entity's Board of Directors or advisory committees; ADCT: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Novartis: Membership on an entity's Board of Directors or advisory committees, Other: Travel funding; Morphosys: Membership on an entity's Board of Directors or advisory committees. Ram:MSD: Honoraria; Gilead: Honoraria; BMS, Takeda, Sanofi, Pfizer: Honoraria; Novartis: Honoraria, Research Funding. Riedell:Novartis: Honoraria; AbbVie, Genmab, ADC Therapeutics, Pharmacyclics, Novartis, BMS, Kite/Gilead, Nurix Therapeutics, Nektar Therapeutics, Takeda, Intellia Therapeutics, Sana Biotechnology, BeiGene, Janssen, CVS Caremark: Consultancy; Kite/Gilead: Speakers Bureau; BMS, Kite/Gilead, Novartis, MorphoSys, CRISPR Therapeutics, Calibr, Xencor, Fate Therapeutics, Tessa Therapeutics.: Research Funding. Popplewell:La Roche: Honoraria; Hoffmann: Honoraria; Seattle Genetics: Consultancy, Honoraria; Novartis: Consultancy; Pfizer: Honoraria. Tiwari:Novartis Healthcare Private Limited: Current Employment. Lim:Novartis: Current Employment, Current equity holder in publicly-traded company. Maier:Novartis Pharma AG: Current Employment. Majdan:Novartis: Current Employment. Pasquini:Kite, a Gilead Company: Honoraria, Research Funding; Novartis: Research Funding; Bristol Myers Squibb: Consultancy, Research Funding; Janssen: Research Funding; Kite Brazil: Honoraria.